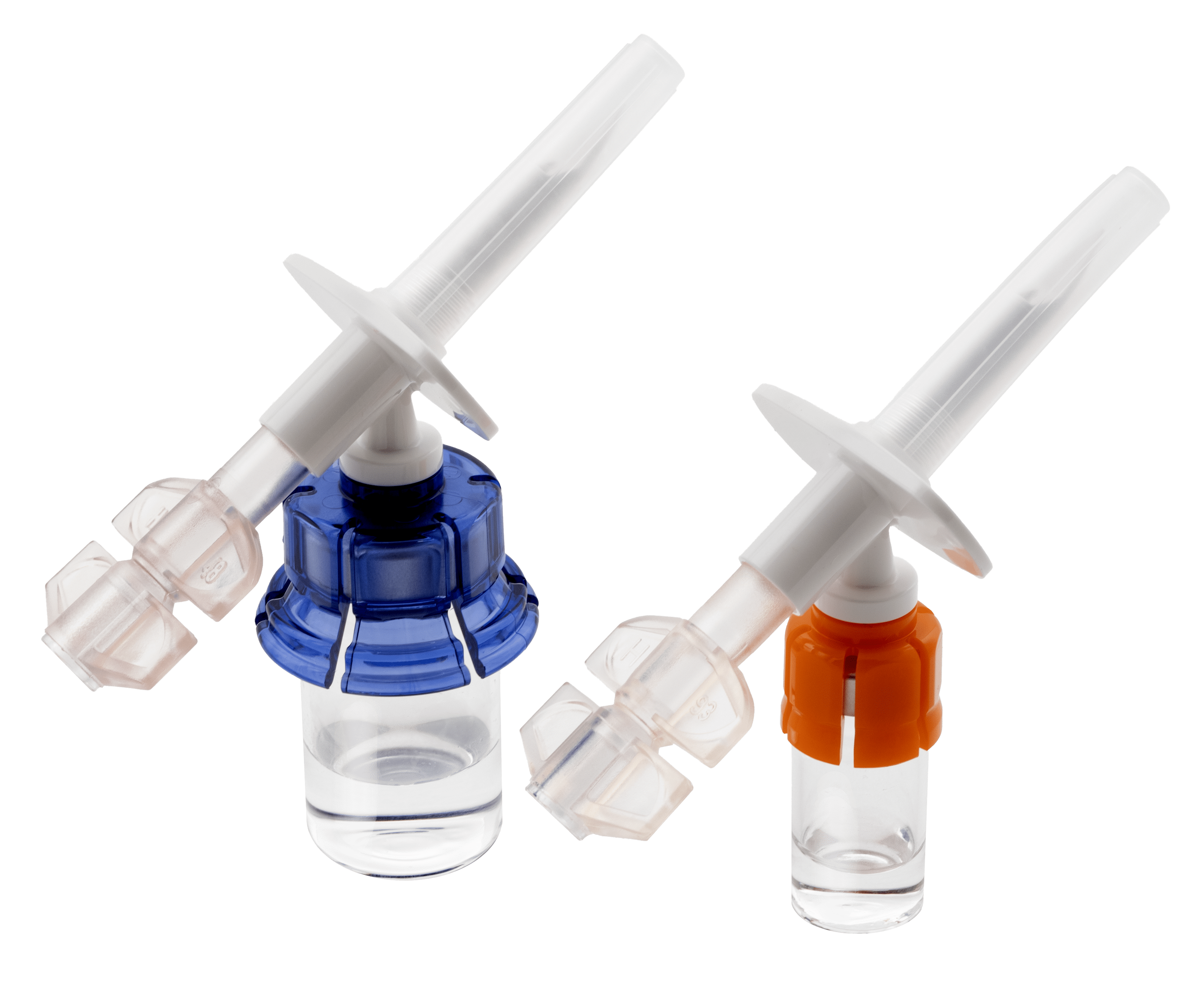
The Vial2Bag Advanced® 13mm and 20mm admixture devices are 510(k) cleared by the United States Food and Drug Administration (FDA). The use of the Vial2Bag Advanced® 13mm and 20mm admixture devices should not be interpreted as modifying, extending, or superseding a drug manufacturer's labeling recommendations for storage and expiration dating, unless otherwise limited by USP <797> compounding standards. Refer to drug manufacturer's labeling and use instructions for recommendations, USP <797>, and applicable institution policy for shelf life and sterility information of reconstituted product and admixture device compatibility. Compatibility of the Vial2Bag Advanced® 13mm and 20mm admixture devices with all drug products has not been confirmed. Do not use the Vial2Bag Advanced® 13mm and 20mm admixture devices with lipids. Failure to follow the instructions provided may result in inadequate medication reconstitution, dilution, and/or transfer, possibly leading to overdose or underdose and/or delay in therapy. Products shown are for INFORMATION purposes only and may not be approved for marketing in specific regions. Please contact your West Pharmaceutical Services, Inc. (West) representative for product availability.
References
1. Tran LK, Anger KE, Dell’Orfano H, Rocchio MA, Szumita PM. Evaluation of Cost, Workflow, and Safety of Implementing a Vial Transfer Device for Ready-to-Mix Drugs at an Academic Medical Center. J Pharm Pract. 2020 Jul 8:897190020938195. doi: 10.1177/0897190020938195. Epubahead of print. PMID: 32638650.
2. van Zanten AR, Engelfriet PM, van Dillen K, et al. Importance of nondrug costs of intravenous antibiotic therapy. Crit Care. 2003; 7(6):R184-R190.
3. Gabay M, Hertig JB, Degnan D, et al. Third consensus development conference on the safety of intravenous drug delivery systems—2018. Am J Health Syst Pharm. 2020;77(3):215-220.
4. Maliekal J, Bertch KE, Witte KW. An update on ready-to-use intravenous delivery systems. Hosp Pharm. 1993;28(10):970–971, 5–7.
5. Hatoum HT, Witte KW, Biedron GT, et al. Microcosting method for small-volume injectables. Am J Hosp Pharm. 1986;43(2): 348-354.
6. Sherrin TP, Miller W, Latiolais CJ. Projecting staffing patterns from time study data in centralized intravenous admixture programs. Am J Hosp Pharm. 1972;29(12):1013-1019.
7. Rosselli D, Rueda JD, Silva MD, et al. Economic evaluation of four drug administration systems in intensive care units in Colombia. Value Health Reg Issues. 2014;5:20-24.
8. Lopez IC, Cuervo MS, Toha AC, et al. Impact of the implementation of vasoactive drug protocols on safety and efficacy in the treatment of critically ill patients. J Clin Pharm Ther. 2016;41(6): 703-710.
9. Sebastian G, Thielke TS. Work analysis of an admixture service. Am J Hosp Pharm
Footnotes
*Extrapolated yearly cost avoidance was measured by multiplying the number of documented yearly administrations by the difference in respective LCSP or RTU costs from V2B system costs.
**This study was conducted using a device that is no longer on the market. West now offers the Vial2Bag Advanced® 13mm and 20mm admixture devices as its transfer device.
***LCSP labor and equipment costs were based on previous time study data that evaluated the costs of preparing small volume injectables (SVI)
**** ISO 8536-4 standard IV spike
*****A drug is considered unused if vial remains unpunctured and within manufacturer's expiration date.
Vial2Bag Advanced® is a registered trademark of West Pharma. Services IL, Ltd., a subsidiary of West Pharmaceutical Services, Inc.